Diamonds and graphite are two forms of what nonmetal?.
Answers
Answer:
Carbon AtomsExplanation: Diamond and graphite are made out of carbon atoms. The difference between the two substance is the arrangement of the carbon atoms inside each one.
Answer:
Diamond and graphite are allotropes of carbon
Explanation:
» Diamond has tetrahedral edges associating through weaker vander waal forces of attraction, it is colorless and glittering, and it is very hard.
» Graphite has hexagonal edges associating vander waal forces, it is black in color and it is soft. Example of graphite is the black rod in a dry cell.
\({}\)
Related Questions
How many mole of HCL is produced if 4g of hydrogen is completely reacted with chlorine
Answers
Answer:
4 moles of HCl are produced.
Explanation:
Given data:
Mass of hydrogen = 4 g
Number of moles of HCl = ?
Solution:
H₂ + Cl₂ → 2HCl
Number of moles of hydrogen:
Number of moles = mass/molar mass
Number of moles = 4 g/2 g/mol
Number of moles = 2 mol
now we will compare the moles of hydrogen and HCl.
H₂ : HCl
1 : 2
2 : 2/1×2 = 4 mol
Thus, 4 moles of HCl are produced.
Is the bond C=O polar or non polar
Answers
Answer:
Non Polar
Explanation:
Non Polar molecule because of its linear symmetric shape
83.20 grams of manganese to moles
Answers
To find the moles present in 83.20 grams of manganese we must use the molar mass of manganese. This mass is equal to 54.94g/mol. So the moles of manganese (Mn) will be:
\(molMn=givengMn\times\frac{1molMn}{MolarMass,gMn}\)\(molMn=83.20gMn\times\frac{1molMn}{54.94gMn}=1.51molMn\)Answer: 83.20 grams of manganese are equivalent to 1.51 moles
A cup of marbles and water is a homogenous mixture / heterogenous mixture. Why?
Answers
Answer:
Another example of a homogeneous mixture is salt water. The rock in Figure above is an example of a heterogeneous mixture. A heterogeneous mixture varies in its composition
Explanation:
Plz help!!! This is timed!!!

Answers
Please correct me if I’m wrong. :)
photoautotrophs are the only organisms that can incorporate (fix) carbon dioxide into biomolecules.
true
false
Answers
False. Photoautotrophs are not the only organisms capable of carbon fixation. Other autotrophs and some heterotrophs can also fix carbon dioxide into biomolecules, and industrial processes also involve carbon fixation.
While photoautotrophs, which are organisms that use light energy and carbon dioxide to produce organic compounds, are capable of carbon fixation, they are not the only organisms that can do so. Other autotrophs, such as chemoautotrophs, which derive energy from inorganic compounds, and some heterotrophs, which obtain energy by consuming other organisms, can also fix carbon dioxide into biomolecules. Additionally, many industrial processes, such as the Haber-Bosch process, involve the fixation of carbon dioxide as a key step. Therefore, while photoautotrophs play an important role in carbon fixation in the biosphere, they are not the only organisms capable of this process.
Learn more about Photoautotrophs here:
https://brainly.com/question/13562991
#SPJ11
Considere uma molécula de glicose (C6H12O6) e indique: a-) O número de átomos de carbono_____________________ b-) O número de átomos de hidrogênio __________________ c-) O número de átomos de oxigênio ___________________ d-) Quantos átomos teremos ao todo se tivermos 8 moléculas dessa substância? _______ e-) Quantos átomos teremos de cada elemento com 20 moléculas de glicose
Answers
Answer:
Considere uma molécula de glicose (C6H12O6) e indique: a-) O número de átomos de carbono = 6 b-) O número de átomos de hidrogênio = 12 c-) O número de átomos de oxigênio = 6d-) Quantos átomos teremos quando descubro se temos 8 moléculas dessa substância?
48 oxigênio, 48 carbono e 96 hidrogênio e-) Quantos átomos de cada elemento teremos com 20 moléculas de glicose?
160 de oxigênio e carbono e 240 de hidrogênio.
Explanation:
A glicose é uma das moléculas mais usadas no organismo humano, pois é uma molécula em que sua decomposição é muito mais fácil que os aminoácidos, portanto, eles produzem como produto a moeda de energia que é ATP mais facilmente do que outros alimentos .
A glicose não é encontrada apenas em alimentos açucarados, isso é um mito, na verdade é a unidade de carboidratos (de assados a doces)
Balance this chemical equation
__K+__H2O>>> __KOH+__H2
Answers
Answer:
2K+2H2O=2KOH+H2
Explanation:
Try to ensure there is same number of reactants as products
At what temperature does ice melt at 101kPa?
Answers
Answer:
Explanation: I also have a question if anyone can help in chem please
The reaction 2NoBr(g) ---> 2NO(g)+Br2(g) is a second order reaction with a rate constant of 0.80M^-1 s^-1 at 11 C. If the initial concentration of NOBr is 0.0440 M, the concentration of NOBr after 12 seconds is _________.
Answers
Answer:
noe sory opwqkfj
s
Explanation:
How do you know a physical change has occurred when evaporating water produces gaseous water?
A
A chemical change has resulted in a new substance.
B
A chemical change has resulted in a change of state.
C
A physical change has resulted in a phase change, not a change in composition.
Answers
Answer:
Its b its the state not anything else
A physical change has occurred has occurred when evaporating water producing gaseous water as a physical change results in a phase change, not a change in composition.
What is a physical change?
Physical changes are defined as changes which affect only the form of a substance but not it's chemical composition. They are used to separate mixtures in to chemical components but cannot be used to separate compounds to simpler compounds.
Physical changes are always reversible using physical means and involve a change in the physical properties.Examples of physical changes include melting,boiling , change in texture, size,color,volume and density.Magnetism, crystallization, formation of alloys are all reversible and hence physical changes.
They involve only rearrangement of atoms and are often characterized to be changes which are reversible.
Learn more about physical changes,here:
https://brainly.com/question/17931044
#SPJ2
I need help on this asp
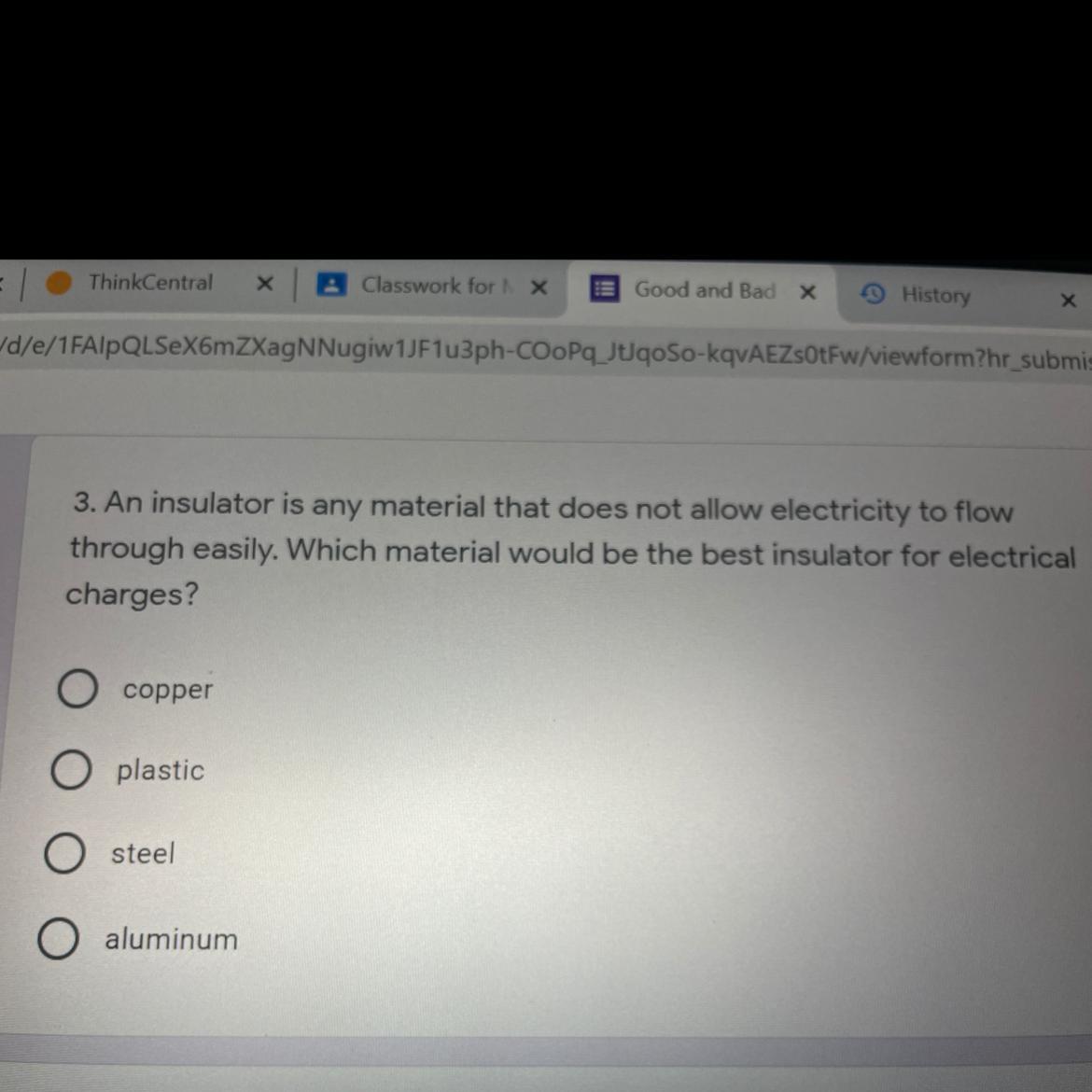
Answers
Answer:
copper or steel
Explanation:
hope this helps
Explanation:
A solution of NaCl was prepared in the following manner: 0.0842 g of NaCl is massed out on an analytical balance. The solid is transferred to a 25.00 mL volumetric flask. Deionized water is added to the flask such that the bottom of the meniscus is at the line. A 1.00 mL aliquot of the stock solution is transferred to a 50.00 mL volumetric flask using a volumetric pipet and diluted to volume. 6. Calculate the concentration of NaCl in the resulting solution in mg/L NaCl. (answer = 67.4 mg/L) 7. Calculate the concentration of NaCl in the resulting solution using propagation of error through the calculation. Use the manufacturer's tolerance values as the absolute error. The tolerances can be found in Chapter 2 of the Harris text. Assume a Class 1 balance and Class A glassware. Treat the tolerances as random error. (answer = 67.4+0.4 mg/L) 8. Identify 2 possible sources of random (indeterminate) error. Identify 2 possible sourses of systematic (determinate) error.
Answers
Two possible sources of systematic (determinate) error in the experiment are; Incorrect calibration of volumetric glasswareIncorrect mass of NaCl
To calculate the concentration of NaCl in the resulting solution in mg/L NaCl, we can use the formula; Concentration (mg/L) = (Mass of solute ÷ Volume of solution in L) × 1000 g / 1 mg NaCl is present in the stock solution of 25 mL. So, the mass of NaCl in the solution would be;0.0842 g ÷ 25 mL = 0.00337 g/mL. Now, in the resulting solution, a 1.00 mL aliquot of the stock solution is transferred to a 50.00 mL volumetric flask and diluted to volume. Therefore, the volume of the resulting solution is 50.00 mL. We will substitute these values in the formula, Concentration (mg/L) = (0.00337 g/mL ÷ 50 mL) × 1000 g / 1 mg concentration (mg/L) = 67.4 mg/L. Therefore, the concentration of NaCl in the resulting solution in mg/L NaCl is 67.4 mg/L.7. Concentration = 67.4 mg/LTolerance = 4.28 mg/LTotal concentration = 67.4 + 4.28 mg/L = 71.68 mg/LWe round off this value to one decimal place; Total concentration = 71.7 mg/LTherefore, the concentration of NaCl in the resulting solution using propagation of error through the calculation is 67.4+0.4 mg/L.8. Two possible sources of random (indeterminate) error in the experiment are; Errors in temperature measurement. Errors in measurement of water volume. Two possible sources of systematic (determinate) error in the experiment are; Incorrect calibration of volumetric glasswareIncorrect mass of NaCl.
Learn more about NaCl
https://brainly.com/question/32275922?
#SPJ11
How much potassium iodide to take for radiation protection.
Answers
Age GroupKI DosageNumber of 65-mg tabletsAdults over 18 years130 mg2Over 12 - 18 years and over 150 pounds130 mg2Over 12 - 18 years and less than 150 pounds65 mg1Over 3 -12 years65 mg1
Explanation:
With the above balanced equation, make at least four mole ratios (one is done for you):
6 KOH
2 K3PO4
Answers
The four mole ratios are: 6 KOH / 2 K₃PO₄, 2 KOH / 1 Co₃(PO₄)₂,
1 K₃PO₄ / 1 Co₃(PO₄)₂, 1 Co(OH)₂ / 2 KOH.
What do you understand by mole ratio?Mole ratio refers to the ratio between the number of moles of two substances in a chemical reaction.
The given chemical equation is:
2 KOH + Co₃(PO₄)₂ → K₃PO₄ + Co(OH)₂
And the first mole ratio given is:
6 KOH / 2 K₃PO₄
To find the other mole ratios, we need to first balance the chemical equation. It is already balanced, so we can proceed to find the other mole ratios:
(2) 2 KOH / 1 Co₃(PO₄)₂
This ratio indicates that two moles of potassium hydroxide react with one mole of cobalt(II) phosphate.
(3) 1 K₃PO₄ / 1 Co₃(PO₄)₂
This ratio indicates that one mole of potassium phosphate is produced for every mole of cobalt(II) phosphate that reacts.
(4) 1 Co(OH)₂ / 2 KOH
This ratio indicates that one mole of cobalt(II) hydroxide is produced for every two moles of potassium hydroxide that react.
Therefore, the four mole ratios are:
(1) 6 KOH / 2 K₃PO₄
(2) 2 KOH / 1 Co₃(PO₄)₂
(3) 1 K₃PO4 / 1 Co₃(PO₄)₂
(4) 1 Co(OH)₂ / 2 KOH
Learn more about moles here:
https://brainly.com/question/26416088
#SPJ1
Correct question is:
KOH + Co₃(PO₄)₂ →
With the above balanced equation, make atleast four mole ratios ( one is done for you ):
\(\frac{6KOH}{2K3PO4}\) - - -
What is the mass of 3.50 moles of Ba(NO2)2?
Answers
Answer:
802.69 g
Explanation:
The molar mass of Barium nitrite is 229.34 g/mol, so 3.5 moles of it will have a mass of ...
3.5 mol × 229.34 g/mol = 802.69 g
predict a likely mode of decay for the following unstable nuclide: rn-196. EXPLAIN why this might be so. a) Mo-109 b) Fr- 202 c) Rn- 196 d) Sb - 132 e) P - 27 f) Ru - 90
Answers
Rn-196 is likely to decay into Mo-109 due to beta decay, as Mo-109 has a higher stability and a lower atomic number, which leads to a more favorable energy state.
Rn-196 is an unstable nuclide, and to achieve a more stable configuration, it is expected to undergo radioactive decay. Among the given options, Mo-109 is the most likely mode of decay for Rn-196. This decay occurs through beta decay, where a neutron in the Rn-196 nucleus transforms into a proton, emitting a beta particle (an electron) in the process.
The resulting nucleus, Mo-109, has a higher stability and a lower atomic number than Rn-196. This transition to a lower atomic number allows the nucleus to achieve a more favorable energy state, leading to increased stability. Therefore, Rn-196 decays into Mo-109 through beta decay.
For more information on decay visit: brainly.com/question/31966018
#SPJ11
#7) How many waves are in this picture?

Answers
Answer:
4
Explanation:
Answer:
B.
Explanation:
I don't know much about this subject, but it seems that a wave is when the line is above the line in the middle.
The volume of a gas is 204 ml when the pressure is 925 kPa at constant temperature what is the final pressure if the volume increases to 306 ml?
Answers
Calculate. 925 x 1.5 is 412.5 + 412.5 + 412.5.
This totals to 1387.5 kPa.
Is climate the same as weather?
Answers
No they are not, as climate is long term conditions for an area while weather is temporary
When the pH of a solution is 2, the concentration of hydronium ions is10-2 M = 0.01 M. Is it acidic or basic?
Answers
Answer:
Explanations:
The formula for calculating the pH of the solution is given as:
\(pH=-log[H_3O^+]\)Given that the pH is 2, then;
\(\begin{gathered} 2=-log[H_3O^+] \\ log[H_3O^+]=-2 \\ [H_3O^+]=10^{-2} \\ [H_3O^+]=0.01M \end{gathered}\)Hence the concentration of the hydronium ion is 0.01M
Since the pH of the solution is less than 7, hence the solution is acidic
A 50. 0 ml sample of gas is cooled from 119° C. If the pressure remains constant, what is the final volume of the gas?
Answers
To use Charles's Law, which states that at constant pressure, the volume of a gas is directly proportional to its temperature. Mathematically, Charles's Law can be expressed as V₁ / T₁ = V₂ / T₂
Where V₁ and T₁ are the initial volume and temperature of the gas, and V₂ and T₂ are the final volume and temperature of the gas, respectively. In this case, we are given that the initial volume (V₁) is 50.0 mL and the initial temperature (T₁) is 119°C. We need to find the final volume (V₂), but we don't have the final temperature (T₂) explicitly mentioned.
However, we are told that the pressure remains constant. When pressure is held constant, the ratio of volumes is directly proportional to the ratio of temperatures. Therefore, we can set up the following equation:
V₁ / T₁ = V₂ / T₂
Plugging in the known values:
50.0 mL / 119°C = V₂ / T₂
Now, we can solve for V₂ by rearranging the equation:
V₂ = (50.0 mL / 119°C) * T₂
Since we don't have the specific final temperature, we cannot calculate the final volume without additional information about the final temperature of the gas.
Learn more about Charles's Law here
https://brainly.com/question/14842720
#SPJ11
How long does it take for the earth to rotate
A. 24hrs
B. 1 yr
C. 2 weeks
D. 5 months
Answers
Answer:
24 hrs it takes for the earth to rotate
Carotenoids are found in high concentrations in yellow and orange leaves or vegetables. Why do these leaves and vegetables appear yellow or orange
Answers
Carotenoids are found in high concentrations in yellow and orange leaves or vegetables. These leaves and vegetables appear yellow or orange because of Reflect and Transmit.
Reflection and Transmission:
Reflection is the change in direction of a wave front at the interface between two different media in such a way that the wave front returns to the medium in which it was originally located. Common examples include reflections of light, sound, and water waves. According to the law of reflection, in specular reflection (e.g. a mirror) the angle at which the wave strikes the surface is equal to the angle at which it is reflected.
Transmission is defined as the process of transmitting light from any substance. Transmission is the movement of electromagnetic waves through any substance.
Example: When UV light hits a material, the electrons in the material's ground state enter a higher electronically excited state. When an electron in a higher state returns to its ground state, it transfers energy in the form of a photon.
Carotenoids
Carotenoids , also called tetraterpenoids, are yellow, orange, and red organic pigments produced by plants and algae, as well as some bacteria and fungi. Carotenoids give pumpkins, carrots, parsnips, corn, tomatoes, canaries, flamingos, salmon, lobster, shrimp, and daffodils their unique colors. All these organisms can produce carotenoids from fats and other basic organic metabolic components.
Learn more about Carotenoids :
https://brainly.com/question/13631581
#SPJ4
What happens to water molecules when they freeze and become solid ice?
Answers
The potential energy of water is decreased when water molecules when they freeze and become solid ice.
Because molecules have energy, they move constantly. Water molecules move more quickly and are essentially in continuous interaction with one another when they are in a liquid state instead of to a solid state.
The potential energy of the liquid decreases as it cools, which causes the molecules to start moving more slowly. The molecules of water bind together at a temperature of about 0°C to produce ice.
Intermolecular hydrogen bonds keep water molecules together, make it a liquid.
Hence, When water molecules freeze and turn into solid ice, their potential energy decreases.
To learn more about Water molecules, Here :
https://brainly.com/question/13436290?referrer=searchResults
#SPJ4
An unknown gas diffuses 0.25 times as fast as He, What is the molecular mass of the
unknown gas?
Answers
The molecular mass of the unknown gas = 64g/mol
The rate of diffusion of a gas is inversely proportional to the square root of its molecular mass. Let the molecular mass of the unknown gas be M. Then, we have:
Rate of diffusion of unknown gas / Rate of diffusion of He = sqrt(MHe / M)
Since the rate of diffusion of the unknown gas is 0.25 times that of He, we can write:
0.25 = sqrt(MHe / M)
Squaring both sides, we get:
0.0625 = MHe / M
M = MHe / 0.0625
The molecular mass of He is approximately 4 g/mol. Substituting this value, we get:
M = 4 / 0.0625
M = 64 g/mol
Therefore, the molecular mass of the unknown gas is approximately 64 g/mol.
Learn more about molecular mass:
https://brainly.com/question/24191825
2H2O + 3HCl ——> 5H2O + Cl
how many moles of H2O are needed to make 3 moles of HCl?
Answers
Answer: 2 moles
Explanation:
We know from the equation that for every 3 moles of HCl consumed, 2 moles of water are consumed (based on the coefficients)
So, the answer is 2 moles.
when you reach the temperature of the what point, you become a liquid?
Answers
Answer:
depends on what the substance is for example iron has a 2,800F melting point
Explanation:
What volume of 0. 200 m KOH must be added to 17. 5 ml of 0. 135 m H3PO4 to reach the third equivalence point?
Answers
Approximately 31.8 mL of 0.200 M KOH must be added to 17.5 mL of 0.135 M H3PO4 to reach the third equivalence point.
To determine the volume of 0.200 M KOH required to reach the third equivalence point when added to 17.5 mL of 0.135 M H3PO4, we need to consider the stoichiometry and the concept of equivalence points in acid-base titrations.
In an acid-base titration, the third equivalence point is reached when the moles of the acid and base are in a 1:3 ratio. Since we have 17.5 mL of 0.135 M H3PO4, we can calculate the moles of H3PO4 by multiplying its concentration (0.135 M) by its volume (17.5 mL) and converting to liters:
Moles of H3PO4 = 0.135 M × 0.0175 L = 0.00236 mol
Since the acid and base react in a 1:3 ratio, we need 3 times the moles of KOH to neutralize the H3PO4. Therefore, the moles of KOH required can be calculated as:
Moles of KOH = 3 × Moles of H3PO4 = 3 × 0.00236 mol = 0.00708 mol
To find the volume of 0.200 M KOH needed, we can rearrange the equation for molarity:
Molarity = Moles of solute / Volume of solution
Volume of KOH = Moles of KOH / Molarity of KOH
Volume of KOH = 0.00708 mol / 0.200 M = 0.0354 L
Finally, we convert the volume to milliliters:
Volume of KOH = 0.0354 L × 1000 mL/L = 35.4 mL
Rounding to the appropriate number of significant figures, we find that approximately 31.8 mL of 0.200 M KOH must be added to 17.5 mL of 0.135 M H3PO4 to reach the third equivalence point.
Learn more about acid-base titrations
brainly.com/question/32083157
#SPJ11
The particles of a substances are arranged in a definite geometric pattern and are constantly vibrating. This substances can be in
Answers
Answer:easy
Explanation: the answer is '/.'/;==-.'/'.;.;pand'.[;';; because,/.;'[][]';;/..,./. hope it helps )