Answers
Answer: The percent dissociation of butanoic acid is 9.8%
Explanation:
\(C_3H_2CO_2H\rightarrow H^+C_3H_2CO_2^-\)
cM 0 0
\(c-c\alpha\) \(c\alpha\) \(c\alpha\)
So dissociation constant will be:
\(K_a=\frac{(c\alpha)^{2}}{c-c\alpha}\)
Give c= 1.4 mM = \(1.4\times 10^{-3}\) and \(\alpha\) = dissociation constant
\(K_a=1.5\times 10^{-5}\)
Putting in the values we get:
\(1.5\times 10^{-5}=\frac{(1.4\times 10^{-3}\times \alpha)^2}{(1.4\times 10^{-3}-1.4\times 10^{-3}\times \alpha)}\)
\((\alpha)=0.098=9.8\%\)
Thus percent dissociation of butanoic acid is 9.8%
Related Questions
I need help finding arm

Answers
Answer:
line 15 is were you can find the a then you look around for the r but as a heads up the m thats supposed to be there was actually inverted into a w in theory it must have been misprinted.
Explanation:
chemists use solution concentration units to
Answers
How many molecules are in 0.0023 moles of CO2?
Answers
Taking into account the definition of Avogadro's Number, 1.38529×10²¹ molecules in 0.0023 mole of CO₂ .
Definition of Avogadro's NumberAvogadro's number is defined as the number of particles (atoms, molecules, ions, electrons) in 12 grams of carbon-12, that is, in one mole of the substance or compound.
Its value is 6.023×10²³ particles per mole and it applies to any substance.
Molecules of CO₂ in this caseConsidering the definition of Avogadro's number, you can apply the following rule of three: If 1 mole of CO₂ contains 6.023×10²³ molecules, 0.0023 mole of CO₂ contains how many molecules?
amount of molecules of CO₂= (0.0023 moles × 6.023×10²³ molecules)÷ 1 mole
amount of molecules of CO₂= 1.38529×10²¹ molecules
Finally, there are 1.38529×10²¹ molecules.
Learn more about Avogadro's Number:
brainly.com/question/11907018
#SPJ1
Share what you have learned about first aid to your family......
please help me with this.
Answers
Answer:
\( \huge\pink{ \mid{ \underline{ \overline{ \tt Answer :- }} \mid}}\)
=> First Aid enables you to assist persons who become injured in the event of an accident or emergency situation until help arrives.
\( \huge\blue{ \mid{ \underline{ \overline{ \tt Situations :- }} \mid}}\)
=> If an accident happens in the workplace, in your home or in a public space, you can use the help of a first aid kit.
=> In situations such as when someone ingests a harmful substance, suffers from a heart attack, a seizure or stroke, is involved in a motor vehicle accident or is caught in a natural disaster, a person trained and knowledgeable in even the very basics of First Aid can be of extreme importance in assisting the injured person(s) until emergency responders arrive.
The irreversible isomerization A
B was carried out in a batch reactor and the following concentration time data were obtained:
Time vs Concentration data in a Batch reactor
t 0 3 5 8 10 12 15 7.5
mol/h 4 2.89 2.25 1.45 1.0 0.65 0.25 0.07
Determine the reaction order,
, and the specific reaction a rate constant, k, using any method of your choice.

Answers
The reaction order and specific reaction rate constant can be determined by performing the kinetics experiment on irreversible polymerization A. Kinetic experiments can be used to investigate the rate and mechanism of chemical reactions. Chemical kinetics is the study of chemical reactions' speed and pathway.
The term "kinetics" refers to the study of reaction rates, which are determined by measuring the concentration of reactants and products as a function of time.Kinetics experiments can be used to determine the reaction rate and order of reaction. A chemical reaction's rate is defined as the change in the concentration of a reactant or product per unit time. The order of a reaction refers to the number of molecules that must react to produce a product. The order of reaction can be determined by measuring the initial rate of the reaction as a function of concentration.Methods for determining the reaction rate order include the initial rate method, the half-life method, and the integrated rate method. The initial rate method determines the reaction order by measuring the initial rate of the reaction at different reactant concentrations. The half-life method determines the reaction order by measuring the time it takes for the reactant concentration to decrease by half.The integrated rate method determines the reaction order by measuring the concentration of the reactant or product at different times.The specific rate constant can be determined by using the Arrhenius equation, which relates the rate constant to the activation energy, temperature, and frequency factor. The frequency factor can be determined by measuring the rate constant at different temperatures.For such more question on polymerization
https://brainly.com/question/1602388
#SPJ8
What is the oxidation state of N in NaNOz?

Answers
The oxidation state of nitrogen (N) in NaNO3 is +5. option B
To determine the oxidation state of nitrogen (N) in sodium nitrate (NaNO3), we need to assign oxidation numbers to each element in the compound.
In NaNO3, we know that the sodium ion (Na+) has a +1 oxidation state because it is an alkali metal. Oxygen (O) typically has an oxidation state of -2 in compounds, and there are three oxygen atoms in NaNO3. Since the compound is neutral, the sum of the oxidation states must be zero.
Let's assume that the oxidation state of nitrogen is x. Therefore, we can set up the equation:
(+1) + x + (-2) * 3 = 0
Simplifying the equation:
+1 + x - 6 = 0
x - 5 = 0
x = +5
Therefore, the oxidation state of nitrogen (N) in NaNO3 is +5.
The oxidation state of an element indicates the number of electrons it has gained or lost in a compound. In this case, the nitrogen atom in NaNO3 has gained five electrons to achieve a stable oxidation state of +5.
It is important to note that oxidation states are formal charges and do not necessarily represent the actual distribution of electrons in a compound. They are assigned based on a set of rules and can be useful in understanding the reactivity and behavior of elements in chemical reactions.
Option B
For more such questions on oxidation state visit:
https://brainly.com/question/25551544
#SPJ8
Which of the following is/are true?
A in each period the atomic numbers increase from
left to right
b The periods are numbered 1 through 7 on the left-
hand side of the periodic table
C Elements in the same period have chemical
properties which are not all similar
d All of the above
Answers
Answer:
Answer is D. All of the above
Describe Muller's Ratchet.
Answers
Answer:
Muller's ratchet is a paradigmatic model in population genetics which describes the fixation of a deleterious mutation in a population of finite size due to an unfortunate stochastic fluctuation.
Explanation:
Answer:
Muller's ratchet is a paradigmatic model in population genetics which describes the fixation of a deleterious mutation in a population of finite size due to an unfortunate stochastic fluctuation.
Explanation:
This is the correct answer took a test and got this correct thank you to the original user above.
How much HNO3 can be made from 25.0 g of NO2 according to the following reaction?
3 NO2 + H2O → 2 HNO3 + NO
Answers
The amount of \(HNO_3\) that can be made from 25.0 g of \(NO_2\) according to the reaction would be 22.68 grams.
Stoichiometric problemsFrom the balanced equation of the reaction:
\(3 NO_2 + H_2O -- > 2 HNO_3 + NO\)
The mole ratio of \(NO_2\) and \(HNO_3\) is 3:2. In other words, for every 3 moles of \(NO_2\) that react, 2 moles of \(HNO_3\) are produced.
If mole = mass/molar mass
25.0 g of \(NO_2\) will be equivalent to (the molar mass of \(NO_2\) is 46 g/mol):
25/46 = 0.54 moles
From, the mole ratio of 3:2, 0.54 moles of \(NO_2\) will require:
2/3 x 0.54 = 0.36 moles of \(HNO_3\)
The molar mass of \(HNO_3\) is 63.01 g/mol.
Mass of 0.36 moles of \(HNO_3\) = 0.36 x 63.01
= 22.68 g
Thus, the mass of \(HNO_3\) that can be made from the reaction is 22.68 grams.
More on stoichiometric problems can be found here: https://brainly.com/question/14465605
#SPJ1
A 1.00 gram sample of which of the following compounds contains the greatest mass of oxygen? And explain.
1. K2O2
2. Na2O
3. BeO
Answers
Answer:
BeO will have the greatest mass composition of oxygen
Explanation:
To solve this problem, we would find the mass composition of oxygen in each of the given sample.
To find this mass composition, let us derive the molecular mass of the given species;
K₂O₂ = 2(39) + 2(16) = 110g/mol
Na₂O = 2(23) + 16 = 62g/mol
BeO = 9 + 16 = 25g/mol
Now; we know that mass of each sample is 1g;
mass composition of O in K₂O₂ = \(\frac{32}{110}\) x 1= 0.29g
mass composition of O in Na₂O = \(\frac{16}{62}\) x 1 = 0.26g
mass composition of O in BeO = \(\frac{16}{25}\) x 1 = 0.64g
We can see that BeO will have the greatest mass composition of oxygen.
The mass composition of oxygen has been highest in BeO. Thus, option 3 is correct.
The molecular mass has been the mass of each element that has been present in the compound. The molecular mass of given compounds have been:
\(\rm K_2O_2\) = 2(mass of K) + 2(mass of O)= 2 (39) + 2 (16)
= 110 g/mol
\(\rm Na_2O\)= 2(mass of N) + mass of O= 2 (23) + 16
= 62 g/mol
BeO = mass of Be + mass of O= 9 + 16
= 23 g/mol
The mass composition of oxygen in 1 gram of the following compounds have been:
Mass composition = \(\rm \dfrac{Mass\;of\;Oxygen}{Molecular\;mass\;of\;compound}\;\times\;weight\;of\;sample\)
\(\rm K_2O_2\) = \(\rm \dfrac{32}{110}\;\times\;1\;gram\)\(\rm K_2O_2\) = 0.29 grams
\(\rm Na_2O\) = \(\rm \dfrac{16}{62}\;\times\;1\;gram\)\(\rm Na_2O\) = 0.26 grams
BeO = \(\rm \dfrac{16}{23}\;\times\;1\;gram\)BeO = 0.64 grams.
The mass composition of oxygen has been highest in BeO. Thus, option 3 is correct.
For more information about the mass composition, refer to the link:
https://brainly.com/question/13531044
The enthalpy of vaporization for methanol is 35.2 kJ/mol. Methanol has a vapor pressure of 1 atm at 64.7 oC. Using the Clausius-Clapeyron equation, what is the vapor pressure for methanol at 55.5 oC? Give your answer in atmospheres, to the third decimal point.
Answers
Answer: 55.5 oC is 0.014 atm (3rd decimal point)
Explanation:
The Clausius-Clapeyron equation is given as:
ln(P2/P1) = -(ΔH_vap/R) * (1/T2 - 1/T1)
where:
P1 = vapor pressure at temperature T1
P2 = vapor pressure at temperature T2
ΔH_vap = enthalpy of vaporization
R = gas constant = 8.314 J/(mol*K)
Converting the enthalpy of vaporization to J/mol:
ΔH_vap = 35.2 kJ/mol = 35,200 J/mol
Converting temperatures to Kelvin:
T1 = 64.7 + 273.15 = 337.85 K
T2 = 55.5 + 273.15 = 328.65 K
Substituting the values into the equation and solving for P2:
ln(P2/1 atm) = -(35,200 J/mol / 8.314 J/(mol*K)) * (1/328.65 K - 1/337.85 K)
ln(P2/1 atm) = -4.231
P2/1 atm = e^(-4.231)
P2 = 0.014 atm
Therefore, the vapor pressure for methanol at 55.5 oC is 0.014 atm, to the third decimal point.
Experiment 4: A chemist mixes aqueous solutions of sodium hydroxide and aluminum chloride in a double-displacement reaction, which forms a white solid precipitate and a clear solution. Write the complete, balanced molecular equation for the reaction. Include physical states.
balanced equation:
Answers
The balanced molecular equation for the reaction between sodium hydroxide (NaOH) and aluminum chloride (\(AlCl_3\)) in aqueous solution can be written as follows: 2NaOH(aq) + 3\(AlCl_3\)(aq) → 3NaCl(aq) + \(Al(OH)_3\)(s)
In this reaction, sodium hydroxide (NaOH) reacts with aluminum chloride (\(AlCl_3\)) to form sodium chloride (NaCl) and aluminum hydroxide (\(Al(OH)_3\)). The coefficients in the balanced equation indicate the stoichiometric ratio between the reactants and products.
The physical states of the substances are indicated by the symbols (aq) for aqueous solutions and (s) for the solid precipitate.
The reaction is a double-displacement reaction, also known as a precipitation reaction. Double-displacement reactions involve the exchange of ions between two compounds, resulting in the formation of a precipitate.
In this case, sodium hydroxide and aluminum chloride react to form sodium chloride and aluminum hydroxide, with aluminum hydroxide being the white solid precipitate.
It's worth noting that the actual reaction might involve hydrated forms of the compounds, such as NaOH·x\(H_2O\) and \(AlCl_3\)·y\(H_2O\). However, for simplicity, these hydrated forms are not included in the balanced equation.
Overall, the balanced equation represents the chemical reaction between sodium hydroxide and aluminum chloride, showing the reactants, products, and their stoichiometric ratios.
For more such question on balanced molecular equation visit:
https://brainly.com/question/11904811
#SPJ8
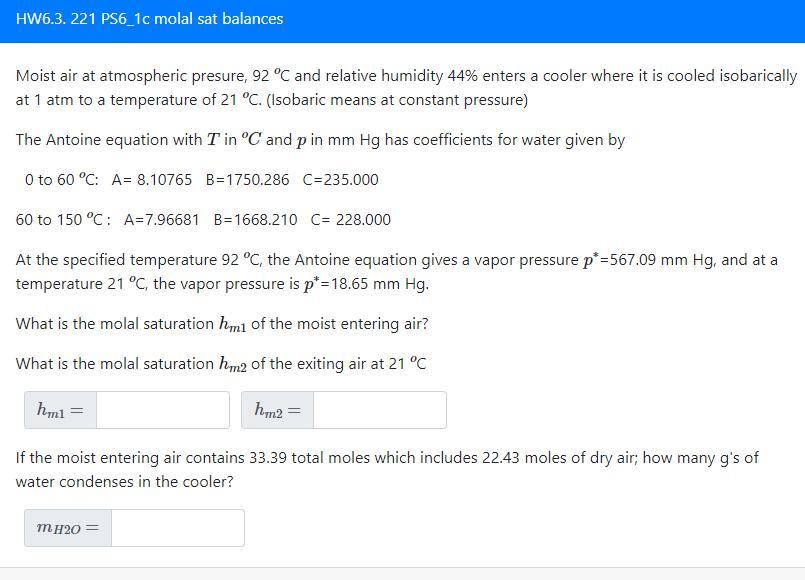
HW6.3. 221 PS6_1c molal sat balances
Moist air at atmospheric presure, 92 C and relative humidity 44% enters a cooler where it is cooled isobarically at 1 atm to a temperature of 21 C. (Isobaric means at constant pressure)
The Antoine equation with T in C and p in mm Hg has coefficients for water given by
0 to 60 C: A= 8.10765 B=1750.286 C=235.000
60 to 150 C : A=7.96681 B=1668.210 C= 228.000
At the specified temperature 92 C, the Antoine equation gives a vapor pressure p*=567.09 mm Hg, and at a temperature 21 C, the vapor pressure is p*=18.65 mm Hg.
What is the molal saturation of the moist entering air?
What is the molal saturation of the exiting air at 21 C
If the moist entering air contains 33.39 total moles which includes 22.43 moles of dry air; how many g's of water condenses in the cooler?
Answers
The molal saturation of the moist entering air is 10.96 mol, molal saturation of the exiting air at 21 C is 0.560 mol and 187.2g of water condenses in the cooler.
Antoine equationThe Antoine equation can be stated as a class of semi-empirical correlations describing the exact relation between vapor pressure and temperature for pure substances.
it is seen that
humidity = 44%
Ptotal = 1 atm = 760 mm Hg
\(P_{H2O}\) = RH/100 X \(P^{*} _{H2O}\) = 52/100 X 567.09
= 294.88 mmHg
Molal saturation = \(\frac{P_{H2O} }{P_{T} -P_{H2O} }\)
= 294.88/ (760 - 294.88)
= 0.6361
Exit air = 100% (after cooling)
\(P_{H2O}\) = \(P^{*} _{H2O}\) at 21°C = 18.65
Hm2 = 18.67/ (760 - 18.65)
= 0.025
Mole of water that enters = mole of total moist - mole of total dry
= 33.39 - 22.43
= 10.96
mole of water exits after cooling
n2 = Hm2 x (mole of dry air)
= 0.025 x 22.43
= 0.560
water condenses = n1 - n2
=10.96 - 0.560
=10.4 mol
=10.4 x 18 g
= 187.2 g
To know more about molality, visit:
https://brainly.com/question/14823885
#SPJ9
A baseball strikes the roof of a car and dents it. The paint on the roof begins to crack and chip, exposing the metal. The exposed metal on the roof rusts, eventually causing a small hole in the roof. 17. Which event is a chemical change? The baseball strikes the roof The roof of the car dents The paint cracks and chips The exposed metal rusts
Answers
Answer:
The exposed metal rusts is an example of a chemical change because rust is an example of a chemical change in objects for example bicycles, scooters, etc.
if a compounds have of calcium oxide is a mass of 5.45 grams, what would be the number of moles for this mass? (round to the 4th decimal place)
Answers
Answer
0.0972
Explanation
Given:
Mass of calcium oxide = 5.45 grams
What to find:
The number of moles for the mass.
Step-by-step solution:
The number of moles in 5.45 grams CaO can be calculated using the mole formula.
\(Mole=\frac{Mass}{Molar\text{ }mass}\)From the periodic table, the molar mass of CaO = (40.078 + 15.999) = 56.077 g/mol
Therefore,
\(Mole=\frac{5.45\text{ }g}{56.077\text{ }g\text{/}mol}=0.0719\text{ }mol\)The number of moles for the mass of 5.45 grams CaO = moles
Suppose that you use a pair of chopsticks and apply a force of 1 N over a distance of 0.01 m. How much work do you do? If the output force of the chopsticks is only 0.5 N, how far do the tips of the chopsticks move?
Answers
Explanation:
Work done = force * perpendicular distance
= 1 * 0.01 = 0.01 joules
If a piece of cadmium with a mass of 37.60 g and a temperature of 100.0 oC is dropped into 25.00 cc of water at 23.0 oC, what will be the final temperature of the system
Answers
Answer:
\(T_{eq}=28.9\°C\)
Explanation:
Hello!
In this case, since it is observed that hot cadmium is placed in cold water, we can infer that the heat released due to the cooling of cadmium is gained by the water and therefore we can write:
\(Q_{Cd}+Q_{W}=0\)
Thus, we insert mass, specific heat and temperatures to obtain:
\(m_{Cd}C_{Cd}(T_{eq}-T_{Cd})+m_{W}C_{W}(T_{eq}-T_{W})=0\)
In such a way, since the specific heat of cadmium and water are respectively 0.232 and 4.184 J/(g °C), we can solve for the equilibrium temperature (the final one) as shown below:
\(T_{eq}=\frac{m_{Cd}C_{Cd}T_{Cd}+m_{W}C_{W}T_{W}}{m_{Cd}C_{Cd}+m_{W}C_{W}}\)
Now, we plug in to obtain:
\(T_{eq}=\frac{37.60g*0.232\frac{J}{g\°C}*100.00\°C+25.00g*4.184\frac{J}{g\°C}*23.0\°C}{37.60g*0.232\frac{J}{g\°C}+25.00g*4.184\frac{J}{g\°C}}\\\\T_{eq}=28.9\°C\)
NOTE: since the density of water is 1g/cc, we infer that 25.00 cc equals 25.00 g.
Best regards!
CuBr2 percent composition
Answers
The percent composition of CuBr₂ is approximately 28.46% of Cu and 71.54% of Br.
To determine the percent composition of CuBr₂ (copper(II) bromide), we need to calculate the mass of each element in the compound and then divide it by the molar mass of the entire compound.
The molar mass of CuBr₂ can be calculated by adding up the atomic masses of copper (Cu) and bromine (Br) in the compound. The atomic masses of Cu and Br are approximately 63.55 g/mol and 79.90 g/mol, respectively.
Molar mass of CuBr₂ = (63.55 g/mol) + 2(79.90 g/mol) = 223.35 g/mol
Now, let's calculate the percent composition of each element in CuBr₂:
Percent composition of copper (Cu):
Mass of Cu = (63.55 g/mol) / 223.35 g/mol × 100% ≈ 28.46%
Percent composition of bromine (Br):
Mass of Br = 2(79.90 g/mol) / 223.35 g/mol × 100% ≈ 71.54%
Therefore, the percent composition of CuBr₂ is approximately:
- Copper (Cu): 28.46%
- Bromine (Br): 71.54%
These values represent the relative mass percentages of copper and bromine in the compound CuBr₂.
for more such questions on composition
https://brainly.com/question/28250237
#SPJ8
How many electrons are being shared

Answers
In the carbon dioxide molecule has four shared pairs of electrons, there will be four covalent bonds created, hence option B is correct.
In organic chemistry, covalent bonds are far more prevalent than ionic ones. Two nuclei are simultaneously drawn to one or more pairs of electrons to form a covalent connection. Bonding electrons are those that are present between the two nuclei.
When atoms share electron pairs, covalent bonding results. Atoms create covalent bonds with one another in order to build a complete electron shell, which increases stability.
Learn more about covalent bonds, here:
https://brainly.com/question/19382448
#SPJ1
You have 165.5 g of Ba(OH)2.
How many moles of barium ions are present in part 1?
_____ mol Ba2+
How many moles of hydroxide ions are present in Part 1?
_____ mol OH1-
Answers
Answer:
moles of Ba²⁺ : 0.97 mol
Moles of OH⁻ : 1.94 mol
Explanation:
Given data:
Mass of Ba(OH)₂ = 165.5 g
Moles of barium ions = ?
Moles of hydroxide ion = ?
Solution:
Number of moles of barium hydroxide:
Number of moles = mass/molar mass
Number of moles = 165.5 g/ 171.34 g/mol
Number of moles = 0.97 mol
one mole of barium hydroxide contain one mole of Ba²⁺ and 2 mole of OH⁻ ion.
Thus in 0.97 moles of barium hydroxide,
moles of Ba²⁺ :
0.97 mol × 1 = 0.97 mol
Moles of OH⁻ :
0.97 mol × 2= 1.94 mol
A student puts some sugar in a glass of water and stirs until the sugar is dissolved.
Why is sugar dissolving in water an example of a physical change?
O because the sugar and water are both clear
B. because the sugar gives off gas bubbles when it is dissolved in the water
O c because the student started with sugar and water, and ended with sugar and water
O D. because the sugar and water molecules break apart, and then recombine in different ways
Answers
Answer:
It does not change the identity of a substance. Sugar if dissolved in water is still sugar.
Explanation:
. Explain with examples following characteristics of chemical reactions:
a. Change of colour b. Evolution of gas c. Change of smell d. Change of state
Answers
Answer:
See explanation.
Explanation:
Hello,
a. In this case, the change of color is evident for instance when copper reacts with nitric acid to form hydrogen and copper (II) nitrate since copper orange-like and nitric acid is colorless, but copper (II) nitrate is green (dry) or blue (hydrated).
b. In this case, when we make react hydrochloric acid and magnesium, we notice a gas giving off while the magnesium chloride remains aqueous, due to the fact that magnesium displaces hydrogen which is given off as a gas.
c. In this case, we can consider an egg since when it is edible it has a tasty smell but when it decomposes to rotten egg, hydrogen sulfide is given off due to the action of specific bacteria, causing a change in smell to a quite stinky one.
d. In this case, a reaction by which a change of state is exhibited is for instance when aqueous lead (II) nitrate reacts with aqueous potassium iodide to yield potassium nitrate which remains aqueous whereas the lead (II) iodide precipitates out as a solid due to its tiny solubility as a yellow solid.
Best regards.
Calculate the number of C atoms in 0.522 mol of C.
Express the number of atoms of carbon numerically.
Nc = ________ atoms of C
Answers
The number of C atoms in 0.522 mol of C is:
Nc = 0.522 mol * Avogadro's number
Nc = 0.522 mol * 6.022 x 10^23 atoms/mol
Nc = 3.145 x 10^23 atoms
So there are 3.145 x 10^23 atoms of C in 0.522 mol of C.
how many formula units of na2so4 are present in a 450 gram sample
Answers
Na₂SO₄ contains 1.90834 X 10²⁴ molecules or formula units in a 450 gram sample.
Formula unit- The chemical formula of an ionic compound that lists the ions in the smallest ratio that corresponds to a neutral electrical charge is known as a formula unit. Chemical formulae are used to describe the constituent parts of a compound in chemistry.
Na₂SO₄ molecular mass is 23x2+ 32+ 16X4.
= 142g/mol
Na₂SO₄ mass = 450g
Na₂SO₄ moles are equal to mass/molar mass.
450g/142g/mol = 3.170 mol.
6.02 X 10²³ mol is the Avogadro's number.
formula unit= moles * Avogadro's number
⇒3.17 X 6.02 X 10²³ molecules= 1.90834 X 10²⁴
Na₂SO₄ therefore contains 1.90834 X 10²⁴ molecules or formula units..
To learn more about formula unit refer- https://brainly.com/question/24529075
#SPJ4
A 1 mL sample weighs 4 g, the density of the sample is
Answers
D=m/v
mass in this case is 4 g
volume is either mL or cm^3 and in this case its 1 mL so
D=4 g/1 mL
D=4 g/mL
Why do you think the mass of the carbon atom shown is 12 atomic mass units (amu)?
Answers
Answer:
The mass of Carbon-12 is exactly 12 amu. But, that is not the number that is reported in your periodic table. Carbon as it exists in nature is not all carbon-12. There is some small amount of carbon-13 and even traces of carbon-14 in there. The periodic table reports the average mass of the carbon as it is found in nature, not that of pure carbon-12. The average mass of carbon is slightly heaver than carbon-12, because of the slight amounts of those heaver isotopes of carbon in nature.
Mark me as brainliest please
Explanation:
A 54.2 g sample of polystyrene, which has a specific heat capacity of 1.880 J-gc, is put into a calorimeter (see sketch at
right) that contains 100.0 g of water. The temperature of the water starts off at 21.0 °C. When the temperature of the water stops
changing it's 34.3 °C. The pressure remains constant at 1 atm.
Calculate the initial temperature of the polystyrene sample. Be sure your answer is rounded to the correct number of significant
digits.
thermometer.
insulated
container
water
sample.
a calorimeter
Answers
Tthe initial temperature of the polystyrene sample is 39.4°C.
Given: Mass of polystyrene sample = 54.2 gSpecific heat of polystyrene = 1.880 J-g°CWater mass = 100.0 g Initial water temperature = 21.0°CWater final temperature = 34.3°CPressure remains constant at 1 atmFormula used:Heat gained by water = heat lost by polystyreneHence,Heat lost by polystyrene = Heat gained by water=> mcΔT = mcΔTwhere,m = mass of polystyrene or waterc = specific heat capacityΔT = change in temperatureThe temperature change is ΔT = 34.3°C - 21.0°C = 13.3°CNow we can use this temperature change to calculate the initial temperature of the polystyrene.Taking the water's specific heat capacity, c = 4.184 J/g°CHeat gained by water = (100.0 g)(4.184 J/g°C)(13.3°C) = 5574 JHeat lost by polystyrene = 5574 JTaking the polystyrene's specific heat capacity, c = 1.880 J/g° ) = 13.3°C Now let's calculate the mass of polystyrene using the specific heat capacity formula.5574 J = (54.2 g)(1.880 J/g°C)(13.3°C - Ti)Ti = 39.4°C
for more questions on polystyrene
https://brainly.com/question/15913091
#SPJ8
The student's lab manual says to mix some of his Na2CO3 solution with an aqueous solution of copper(II) sulfate (CuSO4)
i What evidence of a chemical reaction would he expect to see? Explain your answer.
ii Write a balanced chemical equation to show the reaction. Include state symbols.
iii What kind of reaction is this?
Answers
i When sodium carbonate (Na2CO3) is mixed with an aqueous solution of copper(II) sulfate (CuSO4), the student can expect to see several evidence of a chemical reaction:
Formation of a solid precipitate: When these two solutions are mixed, a solid precipitate of copper(II) carbonate (CuCO3) will form. This is a sign that a chemical reaction has occurred.
Change in color: The reaction between sodium carbonate and copper(II) sulfate will also result in a change in color. The solution may turn a blue or green color, indicating the presence of copper(II) ions.
Release of gases: The reaction between sodium carbonate and copper(II) sulfate may also produce gases, such as carbon dioxide (CO2).
ii The balanced chemical equation for the reaction between sodium carbonate and copper(II) sulfate is:
2Na2CO3(aq) + CuSO4(aq) → 2Na2SO4(aq) + CuCO3(s)
iii This is a double displacement reaction, also known as a metathesis reaction. In this type of reaction, the cations (positively charged ions) and anions (negatively charged ions) of the reactant compounds exchange places to form the products. In this case, the sodium ions (Na+) and the copper ions (Cu2+) exchange places to form sodium sulfate (Na2SO4) and copper carbonate (CuCO3).
How does the Octet Rule apply to molecules?
Answers
The octet rule describes an atom's propensity to favour eight electrons in its valence shell. Atoms with fewer than eight electrons are more likely to interact with one another and create more stable molecules.
How does the octet rule apply to molecules?Based on the observation that the atoms of the main group elements tend to participate in chemical bonding in a way that each atom of the resulting molecule has eight electrons in the valence shell, this theory was developed. Only the core group elements are subject to the octet rule.
What is the best way to implement the octet rule in molecular covalent compounds?Because both atoms receive a full valence shell as a result of the sharing, the octet rule is essential in covalent bonding.
To know more about octet rule visit:-
brainly.com/question/865531
#SPJ1
1. When reactants react to form products, it’s technically called the ____________________________________. But sometimes, there’s an opposite reaction that occurs with the products changing back into the reactants; we call this a ___________________________________.
Answers
A reaction in which reactants react to form products is called forward reaction. In some reactions, products would change back into reactants which is called backward reaction.
In forward reaction, the reactants react with each other and forms product as a result. The reaction occurs from left to right in an equation.
In backward reaction, the products that was formed would revert back to form reactants due to surrounding conditions. The reaction occurs from right to left in an equation.
The type of reaction where both forward and backward reaction takes place is called reversible reaction. When the rate of the reaction between forward and backward reaction becomes equal, it reaches a state of equilibrium.
To know more about Reversible reaction
https://brainly.com/question/16614705
#SPJ1